Let There Be Light
To understand fluorescent lamps, it helps to know a little about light itself. Light is a form of energy that can be released by an atom. It is made up of many small particle-like packets that have energy and momentum but no mass. These particles, called light photons, are the most basic units of light. (For more information, see How Light Works.)
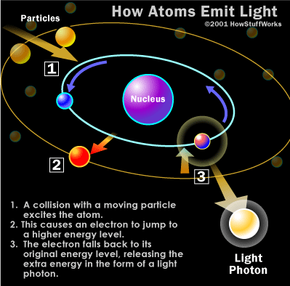
Atoms release light photons when their electrons become excited. If you've read How Atoms Work, then you know electrons are the negatively charged particles that move around an atom's nucleus (which has a net positive charge). An atom's electrons have different levels of energy, depending on several factors, including their speed and distance from the nucleus. Electrons of different energy levels occupy different orbitals. Generally speaking, electrons with greater energy move in orbitals farther away from the nucleus.
Advertisement
When an atom gains or loses energy, the change is expressed by the movement of electrons. When something passes energy on to an atom -- heat, for example -- an electron may be temporarily boosted to a higher orbital (farther away from the nucleus). The electron only holds this position for a tiny fraction of a second; almost immediately, it is drawn back toward the nucleus, to its original orbital. As it returns to its original orbital, the electron releases the extra energy in the form of a photon, in some cases a light photon.
The wavelength of the emitted light depends on how much energy is released, which depends on the particular position of the electron. Consequently, different sorts of atoms will release different sorts of light photons. In other words, the color of the light is determined by what kind of atom is excited.
This is the basic mechanism at work in nearly all light sources. The main difference between these sources is the process of exciting the atoms. In an incandescent light source, such as an ordinary light bulb or gas lamp, atoms are excited by heat; in a light stick, atoms are excited by a chemical reaction. Fluorescent lamps have one of the most elaborate systems for exciting atoms, as we'll see in the next section.