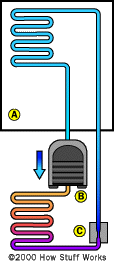
Parts of a Refrigerator
If you pour a little rubbing alcohol on your skin, it'll feel cold -- really cold. It isn't refrigerated, so how does this happen? Well, alcohol evaporates at room temperature the way water evaporates at a low temperature in an oven. As it evaporates, it absorbs the heat on the surface of your skin, making your skin cooler. A special coolant called a refrigerant functions in a refrigerator the way alcohol works on your skin, except in a refrigerator, the coolant is trapped inside a series of coils. As it makes a circuit through them, it changes back and forth from a liquid to a gas.
To pull off this frosty feat, a refrigerator uses five major components:
Advertisement
- Compressor
- Heat-exchanging pipes (serpentine or coiled set of pipes outside the unit)
- Expansion valve
- Heat-exchanging pipes (serpentine or coiled set of pipes inside the unit)
- Refrigerant (liquid that evaporates inside the refrigerator to create the cold temperatures)